Feb 28, 2026

In our previous article, we provided a detailed analysis of anodizing and why it is required for aluminum alloy cylinder tubes.
However, to ensure the best quality of the cylinder tube, we usually perform hard anodizing on it. So what is the difference between hard anodizing and anodizing?
The pretreatment process for hard anodizing is the same as for conventional anodizing, requiring multiple steps such as degreasing, alkaline etching, desmutting, chemical polishing, and cleaning.
Subsequently, the workpiece needs to be immersed in a specialized electrolytic bath. The electrolyte is typically sulfuric acid, oxalic acid, or a mixture of both, but at a significantly lower concentration than that used in conventional anodizing. This is done to slow down the rate of chemical dissolution, which benefits the growth of the oxide film.
While conventional anodizing is carried out at room temperature, hard anodizing must be performed at low temperatures, typically in a 0-5°C environment. This low temperature also serves to inhibit the rate of chemical dissolution. Furthermore, since the oxidation reaction is exothermic, the electrolyte temperature constantly rises during the process. Therefore, the electrolytic bath must be equipped with a refrigeration circulation system to maintain the low temperature.
Additionally, the intensity of the voltage and current greatly influences the rate of the oxidation reaction. Consequently, the voltage and current density used in hard anodizing are much higher than those in conventional anodizing. The voltage is usually increased gradually as the oxidation process progresses. Conventional anodizing typically requires voltages below 20V, whereas hard anodizing can reach up to around 100V.
Finally, there is the oxidation time. Hard anodizing requires a longer duration to achieve a greater film thickness, typically around 1-3 hours. This is roughly three times the time needed for conventional anodizing, and a primary reason for the higher cost of hard anodizing.
After the oxidation is complete, the workpiece must be rinsed with pure water to prevent impurities from entering the honeycomb-like pores of the oxide film. However, hard anodized parts are generally not subjected to color dyeing, as the film layer itself has a dark color, making dyeing ineffective.
Feature | Anodizing | Hard Anodizing |
Film Thickness | 5-25μm | 20-100μm |
Hardness | HV200-350 | HV350-650 |
Pore Size | Larger(10-30nm) | Smaller(5-15nm) |
Texture | Smooth | Fine matte, satin finish |
Color | Transparent | Darker |
Surface Roughness | Minor change | Significantly increased |
Insulation Performance | Good | Excellent |
Corrosion Resistance | Good | Excellent |
Wear Resistance | Good | Excellent |
One reason is that in the low-temperature, low-concentration environment, the electrolytic solution's corrosive effect on the pore wall structure of the aluminum oxide is weakened. This allows for the retention of a dense structure with thick walls and narrow pores, thereby increasing the coating's hardness.
Another key, intrinsic difference is that the conditions of low temperature, high voltage, and extended time catalyze the formation of high-hardness crystalline phases, such as α-Al₂O₃ (Corundum) and γ-Al₂O₃. This can be understood as the "embedding" of numerous tiny, hard crystals within an amorphous matrix—this is the fundamental reason for its ultra-high hardness.
α-Al₂O₃, γ-Al₂O₃, and amorphous Al₂O₃ are three different forms of aluminum oxide. Their differences are analogous to diamond, graphite, and amorphous carbon: they share the same chemical composition, but their atomic arrangement (structure) dictates their vastly different properties.
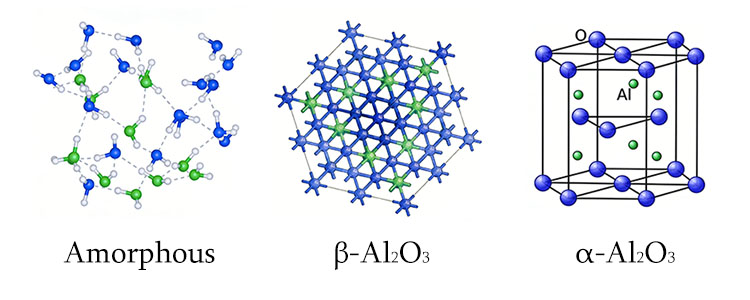
Amorphous Al₂O₃: Abundant in conventional anodized layers. The atomic arrangement is long-range disordered with no regular crystalline structure, resulting in poor density, hardness, and stability.
γ-Al₂O₃: A spinel-type cubic crystal system. The atomic arrangement has some order but contains numerous cation vacancies, making the structure less dense and placing it in a metastable state.
α-Al₂O₃: The most stable hexagonal close-packed crystal system (corundum structure). The atoms are highly ordered and extremely densely packed, bestowing hardness second only to diamond and a thermodynamically stable form capable of withstanding temperatures up to 2000°C and attacks from strong acids and bases.
In aluminum surface treatment, hard anodizing uses low temperature, low concentration, and high voltage to form a dense oxide film of α-Al₂O₃ and γ-Al₂O₃ crystals. Its hardness, wear, and corrosion resistance far exceed conventional anodizing, making it ideal for demanding industrial components like cylinder tubes.
(FK9009)
 Pneumatic Clamping Module Design in Packaging Machinery
Pneumatic Clamping Module Design in Packaging Machinery
 Pneumatic Fittings Replacement Cycle: How Often Should You Change Pneumatic Fittings
Pneumatic Fittings Replacement Cycle: How Often Should You Change Pneumatic Fittings
 Pneumatic Pusher Module Design in Pneumatic Packaging Machines
Pneumatic Pusher Module Design in Pneumatic Packaging Machines
 Vacuum Filter and Response Time: Does Filtration Slow Down Vacuum Systems
Vacuum Filter and Response Time: Does Filtration Slow Down Vacuum Systems
 Metal Seated Pneumatic Ball Valve: When Is Metal Sealing Required?
Metal Seated Pneumatic Ball Valve: When Is Metal Sealing Required?

Feb 15, 2026 Blog
Why do Aluminum Cylinder Barrel Need Anodizing?©1998-2026 Fescolo. A brand of Fokca Automation Co., Ltd. All Rights Reserved Sitemap